Here I explain new data that was presented at the American College of Rheumatology (ACR) 2021 Convergence showing that a sublingual tablet may reduce central sensitization in people with fibromyalgia who have non-restorative sleep.
According to the Institute for ChronicPain central sensitization is a condition of the nervous system that is associated with the development and maintenance of chronic pain. When central sensitization occurs, the nervous system goes through a process called wind-up and gets regulated in a persistent state of high reactivity.
The information presented at the ACR conference was from the Tonixphase 3 RELIEF trial which assessed the efficacy and safety of 5.6 mg TNX-102 SL in patients with fibromyalgia. The study was a double-blind, randomized, placebo-controlled Phase 3 trial with 503 participants.
Sleep disturbances are extremely prevalent in FM, with many studies reporting abnormalities in over 90% of individuals (Chinn, 2016). The current understanding is that pain and abnormal sleep occur in cycles, with the severity of pain correlated with the severity of sleep disturbances. These sleep disturbances include frequent awakenings, non-refreshing sleep, nocturnal restlessness and daytime somnolence. ~ Tonix Pharmaceuticals
Results of the study.
The trial tablet improved sleep, fatigue, and other fibromyalgia symptoms and measures of function according to the study.
In the study TNX-102 SL was associated with:
- decreased daily diary pain scores in patients with fibromyalgia
- 46.8% of patients responding to therapy
- patients reported a ≥30% pain reduction
- Patients more likely to achieve PGIC response than patients on placebo
- improvements in FIQ-R Symptoms, function, sleep quality
- less sleep disturbance
- less fatigue
"...these findings indicate TNX-102 SL primarily targets sleep quality and may thereby reduce central sensitization, leading to improvement at the syndromal level, manifesting as broad-spectrum activity across symptoms of fibromyalgia,”
About TNX-102 SL
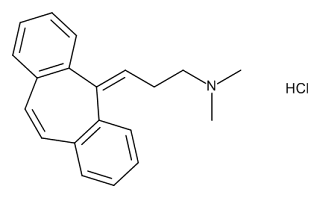
TNX-102 SL is a tablet that is dissolved under the tongue. It is made from cyclobenzaprine hydrochloride and intended to provide broad-spectrum improvement by targeting sleep quality and the stress response.It is in clinical development as a daily bedtime treatment for fibromyalgia, PTSD, alcohol use disorder and agitation in Alzheimer’s disease. The FDA has provisionally accepted the trademark Tonmya® for TNX-102 SL for the treatment of fibromyalgia.
RESOURCES:


Would be wonderful if it works
ReplyDeleteAgreed! We live in hope.
DeletePraying it gets approved and works and most importantly doctors know about it! Too many are still clueless 😥
ReplyDeleteAs of August 15, 2025 the FDA has approved Tonix Pharmaceuticals' TNX-102 SL, under the name Tonmya, for treating adults with fibromyalgia, the first new drug for the indication in over 15 years!!
DeleteFingers crossed
DeleteCan't wait! So Needed 💖
ReplyDeleteFinally approved by FDA in August 2025. WOW!
DeleteI have had fibromyalgia since I was in my early twenties. I haven’t been able to sleep through the night since then and I’m 68 now. It would be heaven to wake up feeling rested after over 40 years.
ReplyDelete